Nexera Amino Acid Analysis System
Nexera Post-column Amino Acid Analysis System
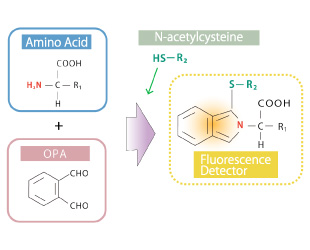
Amino acid analysis occupies an important role in diverse fields such as food science and pharmaceutical manufacturing. Nexera Amino Acid Analysis System uses detection by post-column fluorescence derivatization, with o-phthalaldehyde (OPA)/N-acetylcysteine as the reaction reagents, to selectively and with high sensitivity quantitate the amino acids contained in samples with high levels of contaminants.
Features
-
Selectively detects amino acids based on Shimadzu's highly acclaimed post-column reaction technology and high-sensitivity fluorescence detector.
-
- Superb injection precision over a wide range of volumes
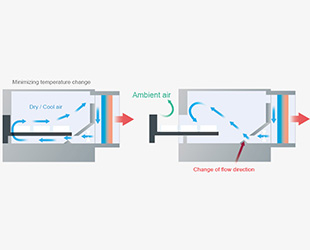
- Unique design to ensure reliable sample cooling -
In terms of the mobile phase, the pH must be carefully prepared in order to obtain separation repeatability in amino acid analysis.
Applications
| Applications | Creation Date |
|---|---|
|
2020-10-27 |
News / Events
-
Shimadzu has released the Nexera MX.
Nexera MX processes twice the number of samples as conventional LCMS systems in the same amount of time.
-
“Advanced i-Series” High Performance Liquid Chromatograph
Retaining the excellent basic functions of the flagship “i-Series”, the Advanced i-Series boasts increased pressure resistance and additional functions to support remote work including working from home.
-
Shimadzu has released the ELSD-LT III Evaporative Light Scattering Detector for HPLC.
The evaporative light scattering detector (ELSD) is a general-purpose universal detector that can even detect components with no UV absorption, such as carbohydrates, lipids, surfactants, and synthetic polymers.
-
Shimadzu has released the Microsampling Device, MSW2™ Type Udck™
For Research Use Only. Not for use in diagnostic procedures.
-
Shimadzu has released Shim-pack MC PLONAS Series.
Shim-pack MC PLONAS 2.7 μm columns reduce the back pressure yet maintain the performance of a sub 2 μm fully porous particle column. The pressure limit is 40 MPa.
-
Application Note: "Analysis of Pharmaceuticals' Impurity - Regulations and Analysis for Carcinogenic Substances -"